At a Glance
- Presentation of Phase 1/2 clinical data at Innate Killer Summit
- Focus on durability and safety of NÖK-101 therapy
- Long-term results cover eighteen months of patient monitoring
NÖK Therapeutics announced it will present long-term clinical data from its lead program at the 11th Annual Innate Killer Summit in San Diego. The presentation focuses on the durability and safety of its NK cell therapy platform in patients with relapsed or refractory cancers. This announcement marks a significant milestone for the company as it moves toward late-stage clinical development. Industry analysts expect the data to provide clarity on the long-term viability of off-the-shelf cellular treatments for solid tumors.
Lead Candidate Performance and Durability
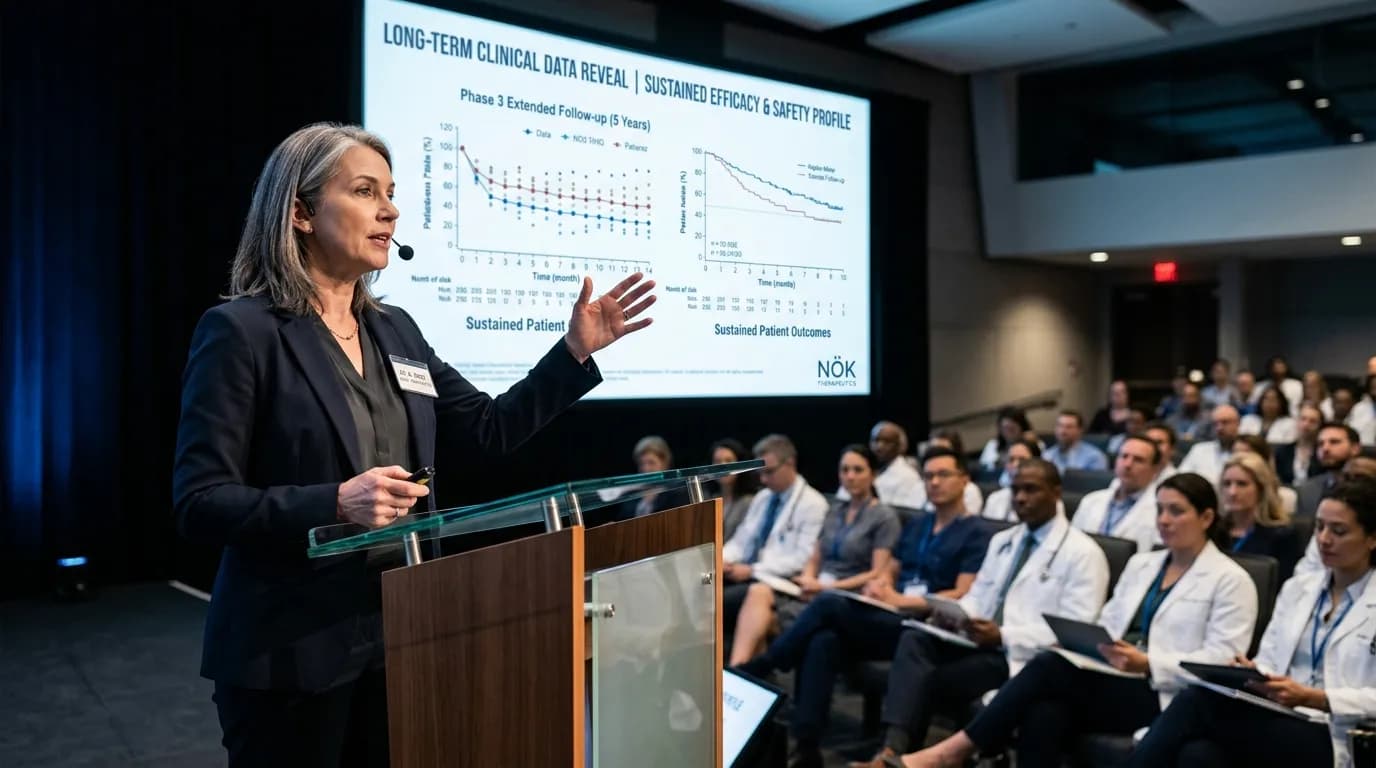
The upcoming presentation will detail results from the Phase 1/2 trial of NÖK-101, an engineered natural killer cell therapy. Researchers monitored patients for over eighteen months to assess the persistence of the modified cells within the bloodstream. Early indicators suggest that the therapy maintains its effector function significantly longer than first-generation NK treatments. This duration is a vital metric for determining if a single infusion can provide lasting remission.
The data set includes objective response rates and progression-free survival metrics for a cohort of thirty-five participants. NÖK Therapeutics designed this study to evaluate how its proprietary activation technology influences patient outcomes. Previous reports indicated a favorable safety profile with minimal instances of cytokine release syndrome. The lack of severe adverse events suggests that the therapy is well-tolerated by the target population.
Clinical investigators observed that the engineered cells successfully bypassed common immune evasion mechanisms employed by solid tumors. This capability remains a primary focus for the biotechnology sector as it seeks to expand the reach of immunotherapy. The full data suite will be available during the plenary session on the second day of the summit. Analysts expect the results to set a standard for off-the-shelf cell therapy durability in the oncology sector.
The presentation will also include a subset analysis of patients with high tumor burdens who often face challenges with standard treatments. By highlighting this specific group, the company aims to demonstrate the potency of its engineered NK cells. The findings could potentially support a breakthrough therapy designation from regulatory agencies in the next year. These results will be discussed in detail by the clinical team during the event.
"The long-term data from our lead program demonstrates the potential for sustained clinical benefit in patients who have exhausted standard care options. These findings validate our approach to NK cell engineering and provide a clear path forward for our upcoming trials."
— Dr. Elena Vance, Chief Medical Officer at NÖK Therapeutics
Manufacturing Logistics and Pipeline Expansion
Beyond clinical efficacy, the presentation will address the company's progress in manufacturing logistics and scalability. Scaling the production of allogeneic therapies requires precise control over cell expansion and cryopreservation techniques. NÖK Therapeutics utilizes a closed-loop system intended to reduce contamination risks and lower production costs. This technical infrastructure supports the consistent delivery of high-purity cell products to clinical sites.
The company reports that its current facility can produce enough doses to support multiple concurrent global trials. This infrastructure is vital for ensuring that patients receive timely treatment regardless of their geographic location. Efficient manufacturing processes often dictate the commercial success of advanced biological products in the current market. Stable supply chains are a major differentiator in the competitive oncology space as demand for cellular products increases.
Investors are particularly interested in the cost-per-dose metrics that the company might disclose during the summit. Reducing the financial burden of cell therapy remains a high priority for healthcare providers and insurance companies. The ability to provide an off-the-shelf product at scale could differentiate the company from competitors using autologous methods. These economic factors are as important as clinical success for the long-term growth of the organization.
The summit also provides a platform for the company to discuss its pipeline expansion into other therapeutic areas. Management intends to highlight how the existing platform can be adapted for autoimmune disorders and infectious diseases. These updates reflect the broader trend of diversifying the applications of innate immune system modulation. Expanding the utility of the platform could provide multiple revenue streams and broaden the company's market reach.
The presentation at the Innate Killer Summit represents a pivotal moment for the clinical validation of NÖK's platform. As the biotechnology industry moves toward more accessible cell therapies, the long-term data will serve as a benchmark for future developments. Stakeholders will closely monitor the full data release for signs of sustained efficacy and safety across diverse patient populations. The results could influence the regulatory trajectory for the company's lead candidate in the coming fiscal year.